"Numerical Simulation of premixed flames in a town gas burner"
![]()

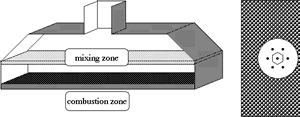
In operation, towngas is blown into a mixing zone. Here, the incoming gas bruises on a plate provided with numerous wholes. Due to this process, the gas is premixed to a homogeneous mixture and leaves with small overpressure through tiny openings of a burner plate into the combustion zone. The arrangement of these openings are schematically presented in the overview of the plate on the righthand side of the picture. The premixing process of the incoming gas takes place at surrounding temperature. Those flow processes were modelled by the group of Prof. Rannacher in Heidelberg as cold and incompressible flow in three dimensions. We were working on the system of the burner plate and the combustion zone. Besides the laminar flow pattern in the combustion zone, predicton of NOx was one of the primary interests of the industry partner. More detailed investigations of nitrogen oxides require the evaluation of a detailed reaction mechanism. The complexity of the given three dimensional problem with detailed chemistry mechanisms makes a numerical simulation with available computer technology unpossible. High temperature ranges, nonlinear coupling of the flow and chemical reaction processes, the wide range of active length scales in the solution are some of the problems concerned. Therfore, detailed investigations of the problem and formulation of a mathematical model had to be done, to reduce the complexity for numerical simulation. Symmetry assumptions, motivated by the structure of the burner plate, the tiny openings and the laminar speed of the incoming gas, led to a two dimensional computational domain given by the shape of a backward facing step. Here the step designes a part of the burner plate, resulting from integration the openings to the computational domain. Simplifications of the complete conservation equations were done for the energy - and species conservation. Here, essential assumptions for reduction of the energy equation are given by Shvab and Zel'dovich. Furthermore, the Lewis number is assumed to be unity. For simplification of species conservation, we assumed the validity of Fick's law of diffusion. For detailed informations see: Bader, G., Eggers. D.,: For discretization of the conservation equations we applied a finite volume method, the so called collocated grid. An essential reason for this decision was it's conservation properties. Alternative discretization methods do not have this properties and imply possible unphysical errors in the solution, especially for simulations of species with low mass fractions like radicals. For the iterative solution of the continuity and momentum equations we applied a stable version of SIMPLE, the SIMPLEC procedure. The energy and species conservation is solved by operator splitting, while the temperature was given by a fixpoint iteration from the enthalpy. In the course of this project, a simulation package on the basis of the object orientated programmers language C++ was implemented. The modules for discretization and solution of isothermal, compressible flow problems, the simulation of one step irreversible reaction mechanisms or detailed reaction mechanisms are interchangeable or expandable. Inheritance of classes was used, to gain a hierarchy for the different model equations. Especially in case of testing various solution methods, for example like ADI, SIP or ILU, this language offers flexibility. In the meanwhile the discretization has been expanded to non-orthogonal grids for the simulation of flow and combustion processes in a rotory piston engine. For computation of the chemical and thermodynamic data, like production and destruction rates, we translated the Fortran chemical reaction library CHEMKIN to C, optimized and extended it's functionality. The following selected results show the influence of different velocities of the incoming gas on the flow and combustion processes. They relate to the simulation of the detailed reaction mechnism including NOx, where the mixture of the incoming gas and it's temperature were held constant.
|